Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Moriyama, Hirotake*
JNC TJ8400 2000-050, 47 Pages, 2000/03
In support of the safety assessment of geologic disposal of high levcl radioactive wastes, the solubility of transuranium elements was studied. The solubility of PuO
 xH
xH O was measured undcr a reducing condition, and the solubility product K
O was measured undcr a reducing condition, and the solubility product K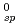 and the stability constant
and the stability constant 
 of Pu(OH)
of Pu(OH) were obtained. The obtained K
were obtained. The obtained K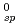 value was found to be much smaller than that predicted by Rai et al. from its dependence on ionic radius. Also, the solubility of PuO
value was found to be much smaller than that predicted by Rai et al. from its dependence on ionic radius. Also, the solubility of PuO 3
3  xH
xH O was measured under an oxidizing condition and the solubility product K
O was measured under an oxidizing condition and the solubility product K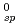 was obtained. In the analysis of hydrolysis constants of actinide ions, it was found that the systematic trend of the hydrolysis constants was well explained by the hard sphere model considering the effective charges of actinide ions.
was obtained. In the analysis of hydrolysis constants of actinide ions, it was found that the systematic trend of the hydrolysis constants was well explained by the hard sphere model considering the effective charges of actinide ions.
J. A. BERRY*; M. BROWNSWORD*; D. J. ILETT*; Linklater, C. M.*; Mason, C.*; TWEED, C. J.*
JNC TJ8400 2000-060, 60 Pages, 2000/02
Batch sorption experiments have been carried out to investigate the sorption behaviour of plutonium onto basalt and sandstone from the appropriate rock-equilibrated waters under different redox eonditions. Redox Potentials in solution were controlled by the addition of two reducing agents and one oxidising agent. Thermodynamic chemical modelling was undertaken to interpret the results. The sorption models were based on iron oxide. They adequately reproduced the data for sorption of plutonium onto sandstone, but tended to underpredict sorption onto basalt.